Chemistry, 21.04.2020 18:07 albattatasraap5wymy
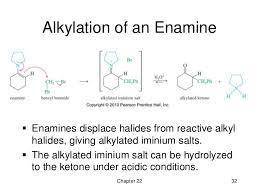
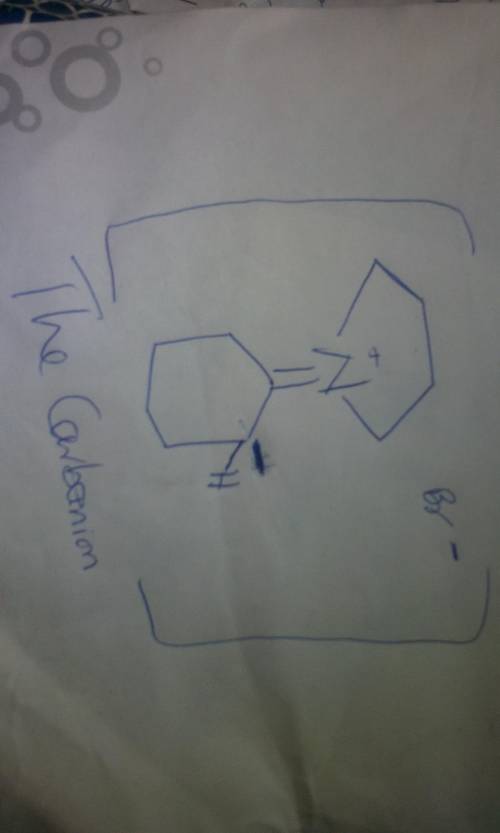
This reaction between an enamine and an alkyl halide involves the following steps: 1. The enamine acts as a nucleophile in an SN2 reaction, displacing bromide ion to form addition product 1; 2. Deprotonation yields carbanion 2; 3. Ring closure leads to the final product. Write the mechanism out on a sheet of paper, and then draw the structure of carbanion 2.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1


You know the right answer?
This reaction between an enamine and an alkyl halide involves the following steps: 1. The enamine ac...
Questions

Mathematics, 27.10.2021 02:50

Biology, 27.10.2021 02:50

Mathematics, 27.10.2021 02:50




History, 27.10.2021 02:50

Mathematics, 27.10.2021 02:50

History, 27.10.2021 03:00


Mathematics, 27.10.2021 03:00




Mathematics, 27.10.2021 03:00




Mathematics, 27.10.2021 03:00

Biology, 27.10.2021 03:00