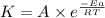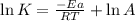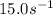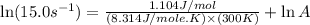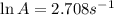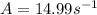A chemical reaction has an energy of activation Ea = 1∙104 J mol-1 at T = 300 K. The first-order rate constant for this reaction was found to be 15.0 s-1. In the presence of a catalyst, the activation energy is reduced to 1∙103 J mol-1. Calculate the pre-exponential factor in the Arrhenius equation

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1


Chemistry, 22.06.2019 21:50
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1

Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3
You know the right answer?
A chemical reaction has an energy of activation Ea = 1∙104 J mol-1 at T = 300 K. The first-order rat...
Questions

Mathematics, 25.11.2020 14:00


Biology, 25.11.2020 14:00


World Languages, 25.11.2020 14:00

Mathematics, 25.11.2020 14:00

French, 25.11.2020 14:00

Mathematics, 25.11.2020 14:00


Mathematics, 25.11.2020 14:00



Biology, 25.11.2020 14:00

World Languages, 25.11.2020 14:00


Biology, 25.11.2020 14:00

Biology, 25.11.2020 14:00


English, 25.11.2020 14:00