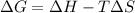Chemistry, 21.04.2020 20:49 pacerskora
G The reaction C(s) + CO2(g) → 2CO(g) is spontaneous only at temperatures in excess of 1100 K. We can conclude that a. ΔG° is negative for all temperatures. b. ΔH° is negative and ΔS° is negative. c. ΔH° is positive and ΔS° is positive. d. ΔH° is negative and ΔS° is positive. e. ΔH° is positive and ΔS° is negative.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
How much heat is released upon converting one mole of steam (18.0 g) from 100.0 ∘c to water at 25.0 ∘c? show work and constants, trying to figure out how it works. only given the heat capacity for steam and water so try to only use that
Answers: 1

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 04:30
What are the three major branches of natural science? • earth and space science, life science, physical science •earth and space science, physical science, chemistry •physical science, life science, chemistry •life science, chemistry, physics
Answers: 1

Chemistry, 22.06.2019 13:10
The last few miles of the marathon are the most difficult for heather, her hair plastered to her head, sweat clinging to her arms, and her legs already feeling as if they had nothing left, just dead weight. after grabbing a cup of ice water, she feels the ice cubes smash against her nose as she gulps some cool refreshment and keeps on running. in these last few miles, the breeze kicks up and she finally feels some coolness against her skin. drips of sweat, once clinging to her forehead, now spill down, and heather feels more pain as the sweat flows into her eyes.which of the following is the most likely reason why the ice struck heather’s nose when she took a drink? a) water can function as a solvent. b) water can store large amounts of heat. c) water can moderate temperatures through evaporative cooling. d) the density of water decreases when it freezes. e) water has a cohesive nature.sweat remained on heather’s forehead and arms because of the a) high salt content of sweat b) cohesive nature of water c) ability of water to moderate heat d) high evaporative cooling effect of water e) ability of water to act as a solvent
Answers: 1
You know the right answer?
G The reaction C(s) + CO2(g) → 2CO(g) is spontaneous only at temperatures in excess of 1100 K. We ca...
Questions

History, 20.11.2020 08:30

Mathematics, 20.11.2020 08:30


Social Studies, 20.11.2020 08:30

Chemistry, 20.11.2020 08:30



SAT, 20.11.2020 08:30

Mathematics, 20.11.2020 08:30

Computers and Technology, 20.11.2020 08:30

English, 20.11.2020 08:30

Mathematics, 20.11.2020 08:30

Mathematics, 20.11.2020 08:30

Computers and Technology, 20.11.2020 08:30


Mathematics, 20.11.2020 08:30

Mathematics, 20.11.2020 08:30

Mathematics, 20.11.2020 08:30


Mathematics, 20.11.2020 08:30