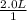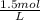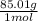Chemistry, 22.04.2020 00:20 venuscagle
1. What mass of the following chemicals is needed to make the solutions indicated?
a. 1.0 liter of a 1.0 M mercury (II) chloride (HgCl2) solution
b. 2.0 liters of a 1.5 M sodium nitrate (NaNO3) solution
c. 5.0 liters of a 0.1 M Ca(OH)2 solution
d. 100 mL of a 0.5 M (NH4)3PO4 solution
2. Calculate the molarity of the following solutions.
a. 12 g of lithium hydroxide (LiOH) in 1.0 L of solution
b. 198 g of barium bromide (BaBr2) in 2.0 L of solution
c. 54 g of calcium sulfide (CaS) in 3.0 L of solution
3. Calculate the volume of each solution, in liters.
a. a 1.0 M solution containing 85 g of silver nitrate (AgNO3)
b. a 0.5 M solution containing 250 g of manganese (II) chloride (MnCl2)
c. a 0.4 M solution containing 290 g of aluminum nitrate (Al(NO3)3)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution? a. 3.88 m, b. 1.03 m, c. 1.5 m, d. 15.5 m
Answers: 3

Chemistry, 22.06.2019 00:00
1) these are barrel shaped microtubules in most animal cells, that organize the spindles during cell division
Answers: 1

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 03:10
Between 2014 and 2016, more than 25,000 children in flint, michigan, drank water that was contaminated with lead from lead pipes. during this time, the city claimed the water was safe to drink. which of these actions could the city have taken to ensure that the drinking water was free from lead?
Answers: 3
You know the right answer?
1. What mass of the following chemicals is needed to make the solutions indicated?
a. 1....
a. 1....
Questions

Mathematics, 12.11.2020 22:50


Chemistry, 12.11.2020 22:50





History, 12.11.2020 22:50

English, 12.11.2020 22:50


Engineering, 12.11.2020 22:50

Arts, 12.11.2020 22:50






Biology, 12.11.2020 22:50