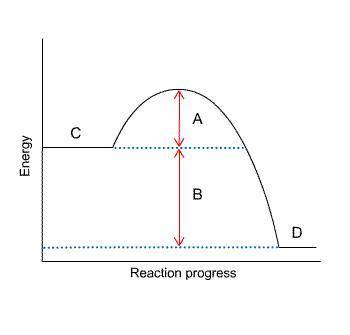
The letter that represents the activation energy from the diagram is letter A.
A chemical reaction occurs only when there is collision between the particles of reactants.
These colliding particles become activated with increased kinetic energy.
Activation energy is the energy barrier that must be overcome before a reaction takes place.
From the diagram,
C is the reactants
D is the products
B is the heat of reaction
While A is the activation energy.
Learn more here:
link