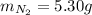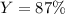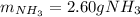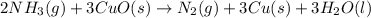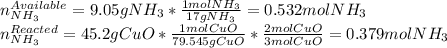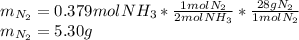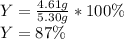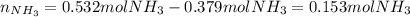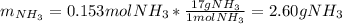Chemistry, 22.04.2020 01:43 sayedabdullah
2 NH3(g) + 3 CuO(s) → N2(g) + 3 Cu(s) + 3 H2O(l) a. What is the limiting reagent when 9.05 g of NH3 reacted with 45.2 g of CuO?(5 points) b. How many grams of N2 can be made?(10 points) c. If 4.61 g of N2 are made, what is the percent yield? (5 points) d. What is the mass of the excess reactant that remains after the reaction. (10 points)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:10
When electrolyzing copper (ll) chloride, what reaction takes place at the anode? what reaction takes place at the cathode?
Answers: 1

Chemistry, 22.06.2019 13:30
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2

Chemistry, 22.06.2019 20:00
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3

Chemistry, 22.06.2019 22:00
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2
You know the right answer?
2 NH3(g) + 3 CuO(s) → N2(g) + 3 Cu(s) + 3 H2O(l) a. What is the limiting reagent when 9.05 g of NH3...
Questions

Mathematics, 08.07.2019 08:30


Mathematics, 08.07.2019 08:30

Mathematics, 08.07.2019 08:30

Health, 08.07.2019 08:30

History, 08.07.2019 08:30

History, 08.07.2019 08:30

Social Studies, 08.07.2019 08:30

Mathematics, 08.07.2019 08:30

English, 08.07.2019 08:30

History, 08.07.2019 08:30

Mathematics, 08.07.2019 08:30

Mathematics, 08.07.2019 08:30


SAT, 08.07.2019 08:30



Mathematics, 08.07.2019 08:30

Biology, 08.07.2019 08:30