Chemistry, 22.04.2020 04:08 destiniout04231
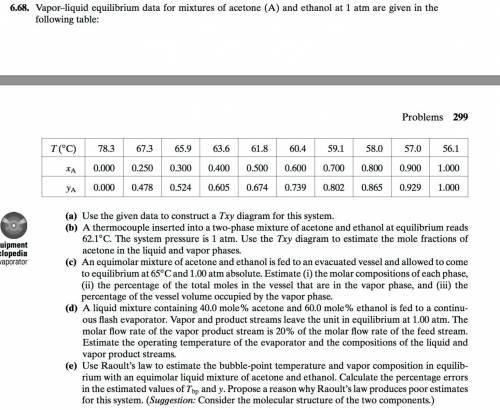
An equimolar mixture of acetone and ethanol is fed to an evacuated vessel and allowedto come to equilibrium at 65°C and 1.00 atm absolute. State a quick way to show thatthe system has two phases. Estimate (i) the molar compositions of each phase, (ii) thepercentage of the total moles in the vessel that are in the vapor phase, and (iii) thepercentage of the vessel volume occupied by the vapor phase.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 21:30
In one or two grammatically correct sentences, write a definition for the term molecule geometry
Answers: 3


Chemistry, 23.06.2019 07:30
Which statement explains which thermometer is more appropriate to measure the temperature of a liquid at 43.6 degrees celsius a) thermometer a, because it measures temperature more accurately than thermometer b b) thermometer b, because it measures temperature more accurately than thermometer a c) thermometer a, because it measures temperature more precisely than thermometer b d) thermometer b, because it measures temperature more precisely than thermometer a
Answers: 2

Chemistry, 23.06.2019 08:10
An experiment is conducted to see if cats preferred skim milk or 2% milk. a cup of skim milkwas put out for 5 kittens and then measured how much the kittens drank over the course of aday. following a cup of 2% milk was purout for the skittens and then masured how much thekittens drank over the course of a day. the same kittens were used and the milk was served atthe same temperature. it was discovered that the cats liked the 2% milk more than the skimmilk. what is the dependent variable in this experiment?
Answers: 1
You know the right answer?
An equimolar mixture of acetone and ethanol is fed to an evacuated vessel and allowedto come to equi...
Questions


Mathematics, 04.07.2019 17:30




English, 04.07.2019 17:30

History, 04.07.2019 17:30

Mathematics, 04.07.2019 17:30

English, 04.07.2019 17:30



Chemistry, 04.07.2019 17:30


Arts, 04.07.2019 17:30



Mathematics, 04.07.2019 17:30


English, 04.07.2019 17:30

Mathematics, 04.07.2019 17:30