Chemistry, 22.04.2020 04:52 alyssamaize
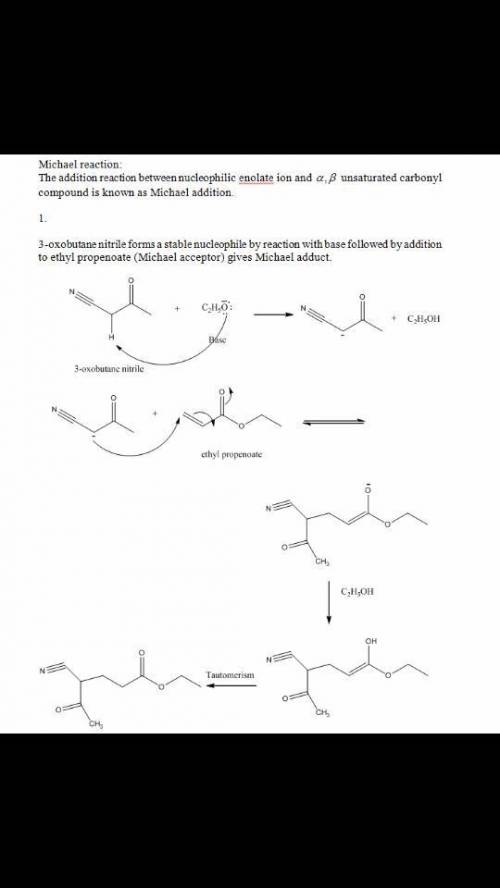
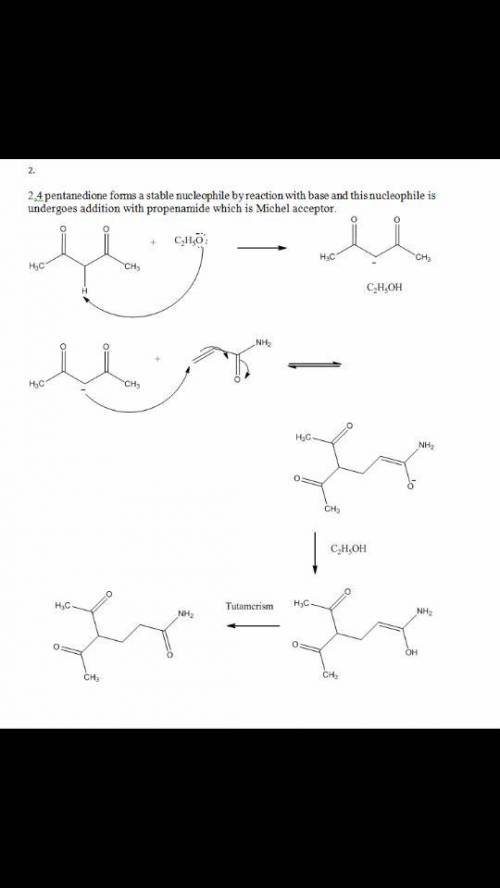
Please help! The Michael reaction is a conjugate addition reaction between a stable nucleophilic enolate ion (the donor) and an α,β-unsaturated carbonyl compound (the acceptor). 1.) Draw the structure of the product of the Michael reaction between ethyl propenoate and 3-oxobutanenitrile.2.)The Michael reaction is a conjugate addition reaction between a stable nucleophilic enolate ion (the donor) and an α,β-unsaturated carbonyl compound (the acceptor). Draw the structure of the product of the Michael reaction between propenamide and 2,4-pentanedione.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3

Chemistry, 23.06.2019 05:00
110 g of water (specific heat = 4.184 j/g c) and 100 g of a metal sample (specific heat = 0.397 j/g c) are heated from 25 degrees c to 75 degrees c. which substance required more thermal energy?
Answers: 1

Chemistry, 23.06.2019 06:40
8. how much enthalpy/heat is transferred when 0.5113gof ammonia (nh3) reacts with excess oxygen according| to the following equation: 4nh3 +502 - 4n0+ 6h20ah = -905.4j
Answers: 1
You know the right answer?
Please help! The Michael reaction is a conjugate addition reaction between a stable nucleophilic eno...
Questions


English, 23.06.2019 23:30


Mathematics, 23.06.2019 23:30

History, 23.06.2019 23:30




Biology, 23.06.2019 23:30

History, 23.06.2019 23:30

Mathematics, 23.06.2019 23:30

Mathematics, 23.06.2019 23:30

History, 23.06.2019 23:30

Geography, 23.06.2019 23:30

Mathematics, 23.06.2019 23:30




Mathematics, 23.06.2019 23:30