
Chemistry, 22.04.2020 19:49 Xavierayala2003
When CO2(g) reacts with H2(g) to form C2H2(g) and H2O(g), 23.3 kJ of energy are absorbed for each mole of CO2(g) that reacts. Write a balanced thermochemical equation for the reaction with an energy term in kJ as part of the equation. Note that the answer box for the energy term is case sensitive. Use the SMALLEST INTEGER coefficients possible and put the energy term in the last box on the appropriate side of the equation. If a box is not needed, leave it blank. 2CO2 + 5H2 + C2H2 + 4H2O + -46.6kJ

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:40
What is the total reduction potential of a cell in which potassium (k) is reduced and copper (cu) is oxidized? a. 2.59 v b. 3.27 v c. -3.27 v d.-2.59 v
Answers: 1

Chemistry, 22.06.2019 00:00
The pressure inside a hydrogen-filled container was 2.10 atm at 21 ? c. what would the pressure be if the container was heated to 92 ? c ?
Answers: 2

Chemistry, 22.06.2019 10:10
What shape would a molecule with two bound groups and two lone pairs have?
Answers: 1

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2
You know the right answer?
When CO2(g) reacts with H2(g) to form C2H2(g) and H2O(g), 23.3 kJ of energy are absorbed for each mo...
Questions




Engineering, 18.12.2020 17:50

French, 18.12.2020 17:50

World Languages, 18.12.2020 17:50

Mathematics, 18.12.2020 17:50


Mathematics, 18.12.2020 17:50

Mathematics, 18.12.2020 17:50

Mathematics, 18.12.2020 17:50





Mathematics, 18.12.2020 17:50

Mathematics, 18.12.2020 17:50



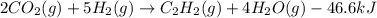
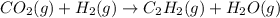
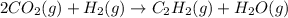
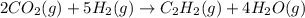
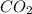 react. So, energy absorbed during the reaction is
react. So, energy absorbed during the reaction is 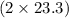 kJ or 46.6 kJ
kJ or 46.6 kJ

