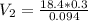Chemistry, 22.04.2020 19:52 blink182lovgabbie
During your reaction, you added 0.3 mL concentrated H2SO4 (18.4 M) as the catalyst. At the end of the reaction, you need to add base to neutralize it. How much volume (in mL) of 10% Na2CO3 to neutralize all the acid present

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 22.06.2019 06:00
How much would the freezing point of water decrease if 4 mol of sugar were added to 1 kg of water(k=1.86 c/mol/kg for water and i=1 for sugar
Answers: 1


Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2
You know the right answer?
During your reaction, you added 0.3 mL concentrated H2SO4 (18.4 M) as the catalyst. At the end of th...
Questions


Biology, 28.01.2020 10:31


Chemistry, 28.01.2020 10:31



Social Studies, 28.01.2020 10:31

Mathematics, 28.01.2020 10:31

Mathematics, 28.01.2020 10:31

Mathematics, 28.01.2020 10:31





Physics, 28.01.2020 10:31


Computers and Technology, 28.01.2020 10:31



Mathematics, 28.01.2020 10:31