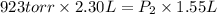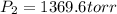A child received a balloon filled with 2.30 L of helium from a vendor at an
amusement park whe...

Chemistry, 22.04.2020 19:54 PrinceBaphomet
A child received a balloon filled with 2.30 L of helium from a vendor at an
amusement park where the pressure in the balloon is 923 torr. What will the final
pressure of the balloon be, in torr, when the child uses some of the helium and
causes the final volume to fall to 1.55 L? Assume that the temperature is held
constant.

Answers: 2
Another question on Chemistry

Chemistry, 23.06.2019 01:30
List and describe the neurological effects of the vocs and other air pollutants,as described by dr.theo colborn
Answers: 2

Chemistry, 23.06.2019 04:31
Which of the following is an example of how telecommunication devices people do their jobs? a.) a security guard checks the time using a digital watch. b.) a banker does some quick math using a solar calculator. c.) a nurse uses a digital thermometer to take a patient’s temperature. d.) a construction worker reports in to his office using a cell phone.
Answers: 1

Chemistry, 23.06.2019 05:00
How many atomic mass units are equal to 1.672×10−24 g of protons?
Answers: 3

Chemistry, 23.06.2019 07:20
F1.5 mol of nabh4 react, how many moles of b2h6 are formed? 2 nabh4(aq) + h2so4(aq) → 2 h2(g) + na2so4(aq) + b2h6(g)
Answers: 1
You know the right answer?
Questions





Mathematics, 12.06.2020 03:57






Mathematics, 12.06.2020 03:57

Mathematics, 12.06.2020 03:57


History, 12.06.2020 03:57






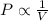
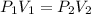
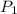 = initial pressure = 923 torr
= initial pressure = 923 torr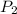 = final pressure = ?
= final pressure = ?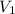 = initial volume = 2.30 L
= initial volume = 2.30 L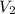 = final volume = 1.55 L
= final volume = 1.55 L