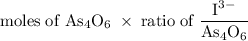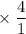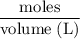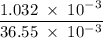Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:00
In the analysis of hair and fiber samples, which does a compound comparison microscope allow for that a conventional compound microscope does not? a. simultaneous observation b. polarization c. fluorescence d. higher magnification
Answers: 2

Chemistry, 21.06.2019 22:00
What is driving behind plate tectonics (plate movment)? a) gravity only b) inertia c) convection and gravity d) the sun theres no option for science so i picked chemistry. plz
Answers: 2

Chemistry, 22.06.2019 10:00
In a water molecule, hydrogen and oxygen are held together by a(an) bond. a) double covalent b) ionic c) nonpolar covalent d) hydrogen e) polar covalent
Answers: 1

Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1
You know the right answer?
A solution of I3¯(aq) can be standardized by using it to titrate As4O6(aq). The titration of 0.1021...
Questions



Mathematics, 18.08.2019 19:00

Mathematics, 18.08.2019 19:00


Social Studies, 18.08.2019 19:00

Social Studies, 18.08.2019 19:00

English, 18.08.2019 19:00


Mathematics, 18.08.2019 19:00


Mathematics, 18.08.2019 19:00

Mathematics, 18.08.2019 19:00

Physics, 18.08.2019 19:00

English, 18.08.2019 19:00


Mathematics, 18.08.2019 19:00

Social Studies, 18.08.2019 19:00

History, 18.08.2019 19:00

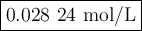
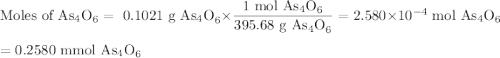
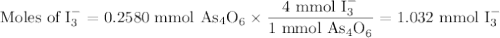
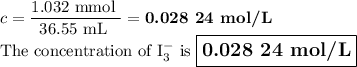
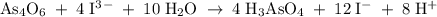
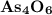 =
= 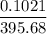
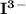 =
=