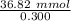Chemistry, 22.04.2020 22:16 dancer4life5642
Buffer capacity is a measure of a buffer solution's resistance to changes in pH as strong acid or base is added. Suppose that you have 125 mL of a buffer that is 0.360 M in both acetic acid ( CH 3 COOH ) and its conjugate base ( CH 3 COO − ) . Calculate the maximum volume of 0.300 M HCl that can be added to the buffer before its buffering capacity is lost.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:30
List four observations that indicate that a chemical reaction may be taking place
Answers: 1



Chemistry, 22.06.2019 23:00
What is the oxidation state of each individual carbon atom in c2o42−?
Answers: 1
You know the right answer?
Buffer capacity is a measure of a buffer solution's resistance to changes in pH as strong acid or ba...
Questions

Mathematics, 16.04.2021 19:30

Mathematics, 16.04.2021 19:30


Mathematics, 16.04.2021 19:30

Mathematics, 16.04.2021 19:30


Health, 16.04.2021 19:30

Biology, 16.04.2021 19:30



Spanish, 16.04.2021 19:30


Biology, 16.04.2021 19:30


Mathematics, 16.04.2021 19:30



Mathematics, 16.04.2021 19:30

Advanced Placement (AP), 16.04.2021 19:30

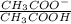
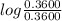
![\frac{[CH_3COO^-]}{[CH_3COOH]} = 0.1](/tpl/images/0619/4300/cb5d0.png)
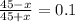
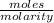 =
=