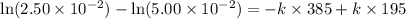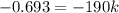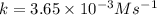Chemistry, 22.04.2020 22:57 grayjasmine46
The reactant concentration in a zero-order reaction was 5.00×10−2 M after 195 s and 2.50×10−2 M after 385 s . What is the rate constant for this reaction? Express your answer with the appropriate units. Indicate the multiplication of units, as necessary, explicitly either with a multiplication dot or a dash.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:20
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2

Chemistry, 22.06.2019 12:30
Clyde and marilyn are riding a roller coaster. during which section(s) of the track is their potential energy converted to kinetic energy? a. from point b to point c only b. from point b to point d only c. from point a to point b only d. from point a to point b and from point c to point d
Answers: 1

Chemistry, 22.06.2019 15:00
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2

Chemistry, 23.06.2019 10:00
Abike ride event is 30 miles. a first aid tent is put at the 3/4 mark of the course. how many miles from the starting point is the first aid tent?
Answers: 1
You know the right answer?
The reactant concentration in a zero-order reaction was 5.00×10−2 M after 195 s and 2.50×10−2 M afte...
Questions



Social Studies, 12.11.2020 06:20

Law, 12.11.2020 06:20

Biology, 12.11.2020 06:20


History, 12.11.2020 06:20



Physics, 12.11.2020 06:20

Social Studies, 12.11.2020 06:20

Mathematics, 12.11.2020 06:20

History, 12.11.2020 06:20


Advanced Placement (AP), 12.11.2020 06:30




History, 12.11.2020 06:30

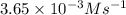
![\ln [A]=-kt+\ln [A_o]](/tpl/images/0619/6365/bdc3f.png)
![[A_o]](/tpl/images/0619/6365/dc622.png) = initial concentration
= initial concentration![[A]](/tpl/images/0619/6365/6aa06.png) = final concentration =
= final concentration = 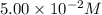 at 195 s
at 195 s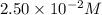 at 385 s
at 385 s![\ln (5.00\times 10^{-2})=-k\times 195+\ln [A_o]](/tpl/images/0619/6365/3a318.png) ............(1)
............(1)![\ln (2.50\times 10^{-2})=-k\times 385+\ln [A_o]](/tpl/images/0619/6365/497e4.png) ............(2)
............(2)![\ln (2.50\times 10^{-2})-\ln (5.00\times 10^{-2})=-k\times 385+\ln [A_o]+k\times 195-\ln [A_o]](/tpl/images/0619/6365/4b64f.png)