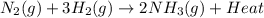Chemistry, 23.04.2020 00:08 shadowsnake
Ammonia is produced commercially by the Haber reaction: N2(8)+ 3H2(8)
The formation of ammonia is favored by
2NH3(8) + heat
an increase in pressure
a decrease in pressure
removal of N2(g)
4.
removal of H2(g)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 20:00
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2

Chemistry, 22.06.2019 21:50
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state.a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
Ammonia is produced commercially by the Haber reaction: N2(8)+ 3H2(8)
The formation of a...
The formation of a...
Questions


Biology, 16.12.2019 20:31




Biology, 16.12.2019 20:31

Computers and Technology, 16.12.2019 20:31









Computers and Technology, 16.12.2019 20:31