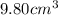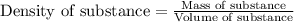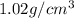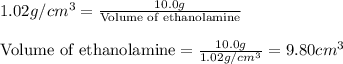Chemistry, 23.04.2020 02:10 abigailweeks10
A chemistry student needs 10.0g of ethanolamine for an experiment. By consulting the CRC Handbook of Chemistry and Physics, the student discovers that the density of ethanolamine is ·1.02gcm−3. Calculate the volume of ethanolamine the student should pour out.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:00
According to the tide table below what time of day will the highest tide occur?
Answers: 1

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
You know the right answer?
A chemistry student needs 10.0g of ethanolamine for an experiment. By consulting the CRC Handbook of...
Questions



Mathematics, 02.02.2020 23:54


Social Studies, 02.02.2020 23:55

Mathematics, 02.02.2020 23:55


Computers and Technology, 02.02.2020 23:55




Social Studies, 02.02.2020 23:55

Mathematics, 02.02.2020 23:55


English, 02.02.2020 23:55

Chemistry, 02.02.2020 23:55

Mathematics, 02.02.2020 23:55


Mathematics, 02.02.2020 23:55