
Chemistry, 23.04.2020 06:41 indiaholmes16
Using the first volume and pressure reading on the table as V1 and P1, solve for the unknown values in the table below. Remember to use the rules of significant figures when entering your numeric response.
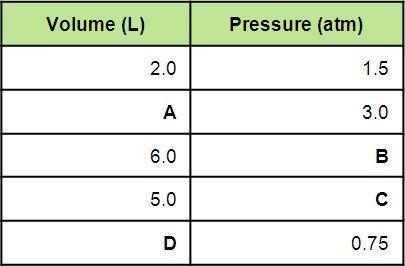

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2


Chemistry, 22.06.2019 19:30
Astring vibrates with a frequency of 10 hz. why can't a person hear the sound waves produced by the vibrating string, no matter how large the amplitude of the waves? out! this is homework and due tomorrow! you so much!
Answers: 2

Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3
You know the right answer?
Using the first volume and pressure reading on the table as V1 and P1, solve for the unknown values...
Questions

Geography, 08.10.2019 08:30

Mathematics, 08.10.2019 08:30

Social Studies, 08.10.2019 08:30





Mathematics, 08.10.2019 08:30



Mathematics, 08.10.2019 08:30



Social Studies, 08.10.2019 08:30

Physics, 08.10.2019 08:30



English, 08.10.2019 08:30

History, 08.10.2019 08:30



