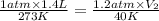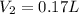Chemistry, 24.04.2020 00:39 soliseric879
A gas has a volume of 1.4 liters at STP. What is the new volume when the temperature is changed
to 40 K and the pressure is changed to 1.2 atm?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Arock can be broken down into different kinds of substances by physical processes. no chemical reactions are needed to separate different parts of a rock into pure substances. this is because a rock is a(n)
Answers: 1

Chemistry, 21.06.2019 16:00
The overall chemical reaction for photosynthesis isshown below: 6co2 + 6h20 → c6h12o6 + 602what mass of glucose (c6h1206) can form from71.89 g co2? (molar mass of c6h1206 = 180.18g/mol; molar mass of co2 = 44.01 g/mol)71.89 g co2=g c6h1206
Answers: 1

Chemistry, 21.06.2019 17:00
Initially, the balloon had 3.0 liters of gas at a pressure of 400 kpa and was at a temperature of 294 k. if the balloon is cooled to 277 k and its volume decreased to 1 l, what will the new pressure in the balloon be?
Answers: 1

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3
You know the right answer?
A gas has a volume of 1.4 liters at STP. What is the new volume when the temperature is changed
Questions

Business, 21.07.2019 22:20

Biology, 21.07.2019 22:20

Health, 21.07.2019 22:20










Social Studies, 21.07.2019 22:20


Computers and Technology, 21.07.2019 22:20

Computers and Technology, 21.07.2019 22:20




Social Studies, 21.07.2019 22:20

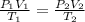
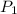 = initial pressure of gas at STP = 1 atm
= initial pressure of gas at STP = 1 atm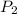 = final pressure of gas = 1.2 atm
= final pressure of gas = 1.2 atm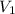 = initial volume of gas at STP = 1.4 L
= initial volume of gas at STP = 1.4 L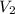 = final volume of gas = ?
= final volume of gas = ?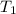 = initial temperature of gas at STP = 273 K
= initial temperature of gas at STP = 273 K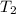 = final temperature of gas = 40 K
= final temperature of gas = 40 K