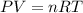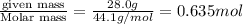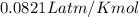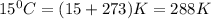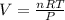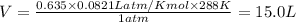Answers: 2
Another question on Chemistry


Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1

Chemistry, 23.06.2019 02:20
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2

Chemistry, 23.06.2019 03:10
Which of the following compounds would be expected to have the strongest ionic bonds? a)the compound that has b)the largest ions with the greatest charge c)the compound that has d)the largest ions with the least charge the compound that has the smallest ions with the greatest charge the compound that has the smallest ions with the least charge
Answers: 2
You know the right answer?
What is the volume of 28.0g of propane at 15C degrees, and 1.2 atm?...
Questions

Biology, 06.07.2019 19:30


History, 06.07.2019 19:30

Spanish, 06.07.2019 19:30

History, 06.07.2019 19:30


Mathematics, 06.07.2019 19:30

History, 06.07.2019 19:30

Health, 06.07.2019 19:30

Chemistry, 06.07.2019 19:30

History, 06.07.2019 19:30

Social Studies, 06.07.2019 19:30

Chemistry, 06.07.2019 19:30


Mathematics, 06.07.2019 19:30



Mathematics, 06.07.2019 19:30


Mathematics, 06.07.2019 19:30