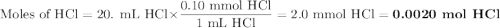Chemistry, 06.05.2020 07:17 gracelong4326
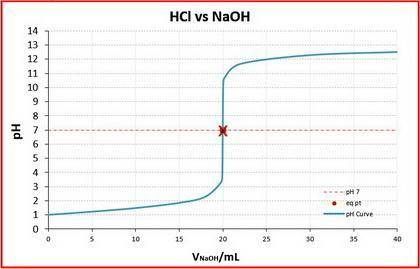
A student was given the task of titrating a 20.mL sample of 0.10MHCl(aq) with 0.10MNaOH(aq) . The HCl(aq) was placed in an Erlenmeyer flask. An equation for the reaction that occurs during the titration is given above.
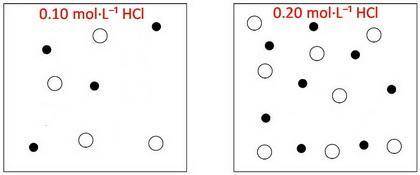
The box below to the left represents ions in a certain volume of 0.10MHCl(aq) . In the box below to the right, draw a representation of ions in the same volume of 0.20MHCl(aq)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Describe the chemical reaction based on the chemical equation below. also, explain whether the equation is balanced.
Answers: 1


Chemistry, 23.06.2019 06:30
Achemist is studying the following equilibirum, which has the given equilibrium constant at a certain temperature: 2 no(g) + cl2(g) < => 2 nocl(g) kp = 2 x 10^(-6)he fills a reaction vessel at this temperature with 13. atm of nitrogen monoxide gas and 12. atm of chlorine gas. use this data to answer the questions: a. can you predict the equilibrium pressure of noci, using only the tools available to you within aleks? y/nb. if you said yes, then enter the equilibrium pressure of nocl at right. round your answer to 1 significant digit.
Answers: 1

Chemistry, 23.06.2019 09:30
What is the best describtion of the side of the moon that faces earth?
Answers: 2
You know the right answer?
A student was given the task of titrating a 20.mL sample of 0.10MHCl(aq) with 0.10MNaOH(aq) . The HC...
Questions

Mathematics, 20.02.2021 20:10

Mathematics, 20.02.2021 20:10


Physics, 20.02.2021 20:10





Chemistry, 20.02.2021 20:10

Chemistry, 20.02.2021 20:10



Chemistry, 20.02.2021 20:10

Computers and Technology, 20.02.2021 20:10

English, 20.02.2021 20:10

Chemistry, 20.02.2021 20:10

Social Studies, 20.02.2021 20:10


Mathematics, 20.02.2021 20:10