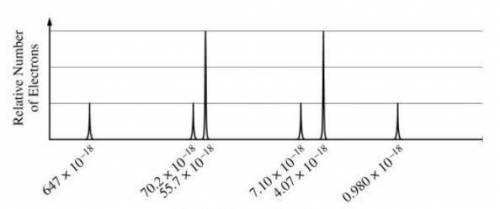
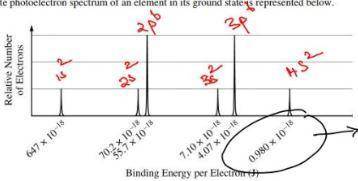
Chemistry, 06.05.2020 05:58 CrsvrBryan7852
A) based on the spectrum, (i) write the ground-state electron configuration of the element, and (ii) identify the element. (b) calculate the wavelength, in meters, of electromagnetic radiation needed to remove an electron from the valence shell of an atom of the element.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 23.06.2019 01:00
Animals that reproduce sexually either do it through external or internal fertilization. read the following statement and decide if it is true or false. birds reproduce through external reproduction which is because the female will then be able to protect the egg.
Answers: 1
You know the right answer?
A) based on the spectrum, (i) write the ground-state electron configuration of the element, and (ii)...
Questions

English, 30.01.2021 06:10



Chemistry, 30.01.2021 06:10

Mathematics, 30.01.2021 06:10

Mathematics, 30.01.2021 06:10

Physics, 30.01.2021 06:10

Mathematics, 30.01.2021 06:10


Mathematics, 30.01.2021 06:10

Law, 30.01.2021 06:10

Mathematics, 30.01.2021 06:10





Mathematics, 30.01.2021 06:10



Mathematics, 30.01.2021 06:10