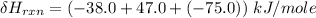Chemistry, 06.05.2020 04:11 addisonrausch
One reaction involved in the conversion of iron ore to the metal is FeO(s) + CO(g) → Fe(s) + CO2(g) Use Hess’s Law to calculate ΔHrxn given the following. 3 Fe2O3(s) + CO(g) → 2 Fe3O4(s) + CO2(g) ΔH = −47.0 k Fe2O3(s) + 3 CO(g) → 2 Fe(s) + 3 CO2(g) ΔH = −25.0 k Fe3O4(s) + CO(g) → 3 FeO(s) + CO2(g) ΔH = 19.0 k

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 03:50
Express the following number in scientific notation. 0.026890 =
Answers: 1


Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2
You know the right answer?
One reaction involved in the conversion of iron ore to the metal is FeO(s) + CO(g) → Fe(s) + CO2(g)...
Questions



Biology, 21.09.2020 01:01

Mathematics, 21.09.2020 01:01
















English, 21.09.2020 01:01

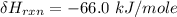
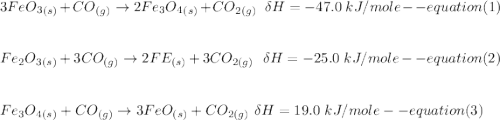
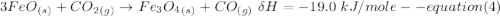
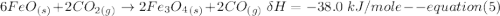
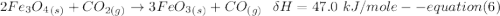
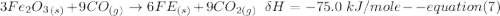

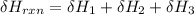 (According to Hess Law)
(According to Hess Law)