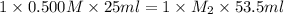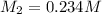Chemistry, 05.02.2020 08:02 sascsl2743
In a titration, 25.0 ml of 0.500 m khp is titrated to the equivalence point with naoh. the final solution volume is 53.5 ml. what is the value of m2 for the reaction?
0.439 m
0.500 m
0.234 m
1.07 m

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
50 pts plz what is the physical state of matter of baking soda.
Answers: 1

Chemistry, 22.06.2019 00:00
For ai it's atomic number is 13 and it's mass number is 27 how many neutrons does it have
Answers: 1

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 07:10
Remember to use the proper number of significant figures and leading zeros in all calculations.gelatin has a density of 1.27 g/cm³. if you have a blob of gelatin dessert that fills a 2.0 liter bottle, what is its mass? 2540 g2500 g3.9 x 10-43.937x 10-4
Answers: 3
You know the right answer?
In a titration, 25.0 ml of 0.500 m khp is titrated to the equivalence point with naoh. the final sol...
Questions

History, 28.02.2021 03:20

Mathematics, 28.02.2021 03:20

English, 28.02.2021 03:20


Mathematics, 28.02.2021 03:20

Mathematics, 28.02.2021 03:20





Mathematics, 28.02.2021 03:20

English, 28.02.2021 03:20



Mathematics, 28.02.2021 03:20

History, 28.02.2021 03:20




Chemistry, 28.02.2021 03:20

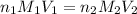
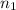 = basicity of an acid = 1
= basicity of an acid = 1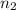 = acidity of a base = 1
= acidity of a base = 1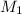 = concentration of KHP = 0.500 M
= concentration of KHP = 0.500 M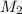 = concentration of NaOH = ?
= concentration of NaOH = ?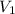 = volume of KHP = 25 ml
= volume of KHP = 25 ml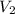 = volume of NaOH = 53.5 ml
= volume of NaOH = 53.5 ml