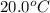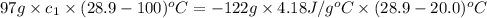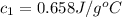Chemistry, 06.05.2020 00:59 faithyholcomb
You are trying to determine the specific heat of a metal. You heat the 97 g piece of metal to 100 °C and place it in a calorimeter with 122 g of water. The temperature of the water increases from 20.0 °C to 28.9 °C. What is the specific heat of the metal?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 23.06.2019 00:50
The chemical formula for emerald is be3al2(sio3)6.an emerald can be decided as
Answers: 3

Chemistry, 23.06.2019 01:00
You wish to prepare a buffer consisting of acetic acid and sodium acetate with a total acetic acetate plus acetate concentration of 250 mm and a ph of 5. what concentrations of acetic acid and sodium acetate should you use
Answers: 1
You know the right answer?
You are trying to determine the specific heat of a metal. You heat the 97 g piece of metal to 100 °C...
Questions

English, 10.11.2020 08:50

History, 10.11.2020 08:50



Mathematics, 10.11.2020 08:50


English, 10.11.2020 08:50

English, 10.11.2020 08:50

English, 10.11.2020 08:50


Biology, 10.11.2020 08:50



History, 10.11.2020 08:50

Chemistry, 10.11.2020 08:50

Mathematics, 10.11.2020 08:50



Mathematics, 10.11.2020 08:50

Arts, 10.11.2020 08:50

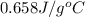
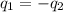
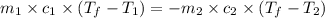
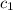 = specific heat of metal = ?
= specific heat of metal = ?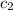 = specific heat of water =
= specific heat of water = 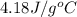
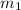 = mass of metal = 97 g
= mass of metal = 97 g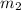 = mass of water = 122 g
= mass of water = 122 g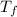 = final temperature of mixture =
= final temperature of mixture = 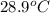
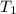 = initial temperature of metal =
= initial temperature of metal = 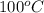
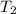 = initial temperature of water =
= initial temperature of water =