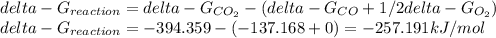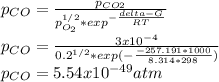Chemistry, 06.05.2020 00:21 Jenniferojeda2002
Hat is the pressure of CO(g) in equilibrium with the CO2(g) and O2(g) in the atmosphere at 25 C? The partial pressure of O2(g) is 0.2 bar and the partial pressure of CO2(g) is 3 * 10-4 bar. CO is extremely poisonous because it forms a very strong complex with hemoglobin. Should you worry?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 23.06.2019 00:30
Nuclear decay is the spontaneous decay of one element into a. an x-ray b. a ray of light c. another element
Answers: 1

Chemistry, 23.06.2019 04:00
How many liters of water can be produced from 5.0liters of butane gas at stp, assuming excess oxygen? c4h10(g) + 02(g) → co2 (e) + h2o (g)
Answers: 2

Chemistry, 23.06.2019 11:00
The lab procedure involves several factors, listed below some were variable and some were constant. label each factor below v for variable ot c for constant
Answers: 1
You know the right answer?
Hat is the pressure of CO(g) in equilibrium with the CO2(g) and O2(g) in the atmosphere at 25 C? The...
Questions

Computers and Technology, 13.02.2020 21:42

Computers and Technology, 13.02.2020 21:42






Mathematics, 13.02.2020 21:42


Physics, 13.02.2020 21:42





Mathematics, 13.02.2020 21:42