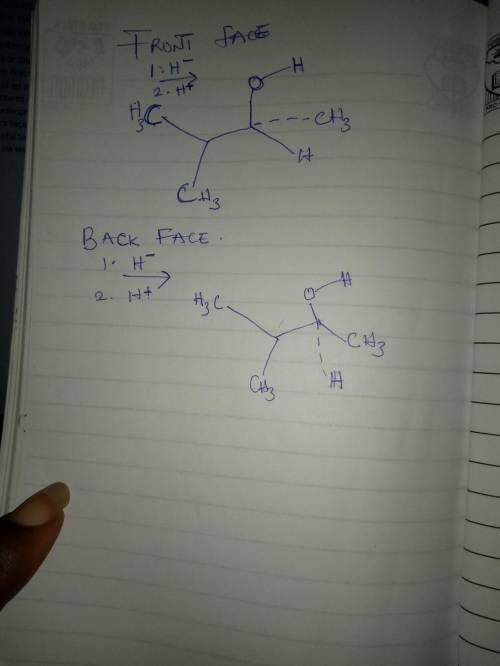
The nucleophilic addition reaction depicted below involves a prochiral ketone carbon atom reacting with a nucleophilic hydride ion source (LiAlH4 or NaBH4) and, subsequently, a proton source (e. g., H2O or dilute aq. HCl). Consequently, the reaction produces a racemic mixture of an alcohol. Finish drawing the structures of the products resulting from nucleophilic attack upon the front and back faces of the carbonyl group, being careful to specify the stereochemistry via wedge-and-dash bonds.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 22.06.2019 15:00
According to the diagram, what sources contribute to the phosphorus found in soil? according to the diagram, phosphorus found in soil contributes phosphorus to what other sources?
Answers: 1

Chemistry, 22.06.2019 21:00
Need what is special about water as a compound? how does water regulate climate? what drives water evaporation? why is the water vapor fresh water when it rises from the ocean? why might freshwater in the form of snow take longer to enter the water cycle again than liquid precipitation? what is an aquifer? what role do people play in the water cycle? plz just answer as many as you can ! thx if you !
Answers: 1
You know the right answer?
The nucleophilic addition reaction depicted below involves a prochiral ketone carbon atom reacting w...
Questions

Mathematics, 13.04.2021 02:30


Mathematics, 13.04.2021 02:30









Geography, 13.04.2021 02:30





History, 13.04.2021 02:30

Social Studies, 13.04.2021 02:30