
Chemistry, 05.05.2020 19:24 elpancho755
Balance the equation by inserting coefficients as needed. equation: ZnS + HBr -> ZnBr_{2} + H_{2}S ZnS + HBr ⟶ ZnBr 2 + H 2 S

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 15:50
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1
You know the right answer?
Balance the equation by inserting coefficients as needed. equation: ZnS + HBr -> ZnBr_{2} + H_{2}...
Questions





Biology, 03.12.2020 20:10

Geography, 03.12.2020 20:10

Mathematics, 03.12.2020 20:10

Physics, 03.12.2020 20:10

Biology, 03.12.2020 20:10





English, 03.12.2020 20:10

Biology, 03.12.2020 20:10





Chemistry, 03.12.2020 20:10

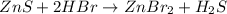
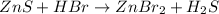
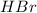 and we get the balanced chemical equation.
and we get the balanced chemical equation.

