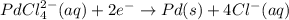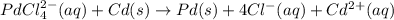Chemistry, 05.05.2020 18:31 cdradlet2001
A voltaic cell that uses the reaction PdCl42−(aq)+Cd(s) → Pd(s)+4Cl−(aq)+Cd2+(aq) has a measured standard cell potential of +1.03 V. You may want to reference (Pages 860 - 867) Section 20.4 while completing this problem. Part A Write the half-cell reaction at the cathode.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:40
What is the study of how matter and energy interact? a. biology b. physics c. planetary science d. chemistry
Answers: 1

Chemistry, 21.06.2019 21:10
Harvey mixes two liquids. which observation of the new mixture most likely indicates a precipitate is forming?
Answers: 2

Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1
You know the right answer?
A voltaic cell that uses the reaction PdCl42−(aq)+Cd(s) → Pd(s)+4Cl−(aq)+Cd2+(aq) has a measured sta...
Questions

English, 05.10.2019 21:30

Chemistry, 05.10.2019 21:30



Mathematics, 05.10.2019 21:30


History, 05.10.2019 21:30

Physics, 05.10.2019 21:30



Health, 05.10.2019 21:30

Mathematics, 05.10.2019 21:30


English, 05.10.2019 21:30

Biology, 05.10.2019 21:30

Mathematics, 05.10.2019 21:30

Geography, 05.10.2019 21:30

History, 05.10.2019 21:30