
Chemistry, 05.05.2020 16:30 hhhhhh8897
21-B. Mn was used as an internal standard for measuring Fe by atomic absorption. A standard mixture containing 2.00 mg Mn/mL and 2.50 mg Fe/mL gave a quotient (Fe signal/Mn signal) 5 1.05/1.00. A mixture with a volume of 6.00 mL was prepared by mixing 5.00 mL of unknown Fe solution with 1.00 mL containing 13.5 mg Mn/mL. The absorbance of this mixture at the Mn wave- length was 0.128, and the absorbance at the Fe wavelength was 0.185. Find the molarity of the unknown Fe solution.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1

Chemistry, 22.06.2019 07:30
Plz mark brainliest 30 points 1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s. 2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1

Chemistry, 22.06.2019 22:00
What mass of glucose is produced when 54g of water react with carbon dioxide
Answers: 1
You know the right answer?
21-B. Mn was used as an internal standard for measuring Fe by atomic absorption. A standard mixture...
Questions



Chemistry, 28.01.2020 04:31


Computers and Technology, 28.01.2020 04:31















Computers and Technology, 28.01.2020 04:31

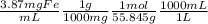 = 0.0693M Fe
= 0.0693M Fe

