
A student is asked to standardize a solution of barium hydroxide. He weighs out 0.978 g potassium hydrogen phthalate (KHC8H4O4, treat this as a monoprotic acid). It requires 35.8 mL of barium hydroxide to reach the endpoint. A. What is the molarity of the barium hydroxide solution? M This barium hydroxide solution is then used to titrate an unknown solution of hydrochloric acid. B. If 17.1 mL of the barium hydroxide solution is required to neutralize 18.6 mL of hydrochloric acid, what is the molarity of the hydrochloric acid solution? M

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:20
Concerning the 10.0 ml of 0.50 m nacl to 100 ml of solution: when a solution is diluted, does it change the number of moles dissolved?
Answers: 3

Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

Chemistry, 23.06.2019 11:30
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 23.06.2019 11:30
How do you calculate the mass of a product when the amounts of more than one reactant are given?
Answers: 3
You know the right answer?
A student is asked to standardize a solution of barium hydroxide. He weighs out 0.978 g potassium hy...
Questions


Mathematics, 28.06.2019 18:30

History, 28.06.2019 18:30

History, 28.06.2019 18:30


Mathematics, 28.06.2019 18:30




Mathematics, 28.06.2019 18:30

History, 28.06.2019 18:30

Mathematics, 28.06.2019 18:30

History, 28.06.2019 18:30



Mathematics, 28.06.2019 18:30



Physics, 28.06.2019 18:30

 = 6.64 mmol Ba(OH)₂
= 6.64 mmol Ba(OH)₂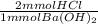 = 4.41 mmol HCl
= 4.41 mmol HCl

