
Consider the two reduction half-reactions: Na+(aq) + e− LaTeX: \longrightarrow⟶ Na(s) Eo = −2.71 V Cl2(g) + 2 e− LaTeX: \longrightarrow⟶ 2 Cl−(aq) Eo = 1.36 VUse the electrode potentials above to calculate Eocell and ∆Gorxn for the reaction below, and determine if it is the reaction for a voltaic cell or an electrolytic cell. 2 Na+(aq) + 2 Cl−(aq) LaTeX: \longrightarrow⟶2 Na(s) + Cl2(g)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:00
What effect does a decrease in temperature have on the overall rate of a chemical reaction? a decrease in temperature decreases . the reaction rate will
Answers: 1

Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2

You know the right answer?
Consider the two reduction half-reactions: Na+(aq) + e− LaTeX: \longrightarrow⟶ Na(s) Eo = −2.71 V C...
Questions



English, 29.04.2021 06:10

English, 29.04.2021 06:10


Mathematics, 29.04.2021 06:10

Mathematics, 29.04.2021 06:10

Mathematics, 29.04.2021 06:10

Geography, 29.04.2021 06:10

Business, 29.04.2021 06:10

Mathematics, 29.04.2021 06:10




English, 29.04.2021 06:10

History, 29.04.2021 06:10

Mathematics, 29.04.2021 06:10

Mathematics, 29.04.2021 06:10

Social Studies, 29.04.2021 06:10

Mathematics, 29.04.2021 06:10

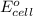 = -4.07v
= -4.07v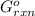 = +785.510 KJ
= +785.510 KJ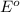 and Δ
and Δ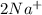 +
+ 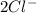 →
→ 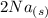 +
+ 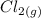
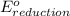 - Δ
- Δ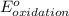 = -2.71v - 1.36v = -4.07V
= -2.71v - 1.36v = -4.07V

