A mixture of helium, nitrogen, and oxygen has a total pressure of 752 mm Hg. The
partial press...

Chemistry, 05.05.2020 05:42 tylorroundy
A mixture of helium, nitrogen, and oxygen has a total pressure of 752 mm Hg. The
partial pressures of helium and nitrogen are 234 mm Hg and 197 mm Hg, respectively.
What is the partial pressure of oxygen in the mixture?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Naoki's bicycle has a mass of 10 kg. if naoki sits on her bicycle and starts pedaling with a force of 168 n, causing an acceleration of 2.8 m/s2, what is naoki's mass?
Answers: 1

Chemistry, 22.06.2019 15:30
The gulf stream is a warm water current that flows away from the equator to northern europe. witch of these does it cause. a. crashes of warm and cool water in the ocean b.colder climates near the equator c.large waves on the cost of europe d.warm climates in northern europe
Answers: 1

Chemistry, 23.06.2019 10:40
Question 17 hydrogen is manufactured on an industrial scale by this sequence of reactions: +ch4gh2og ⇌ +cog3h2g k1 +cogh2og ⇌ +co2gh2g k2 the net reaction is: +ch4g2h2og ⇌ +co2g4h2g k write an equation that gives the overall equilibrium constant k in terms of the equilibrium constants k1 and k2. if you need to include any physical constants, be sure you use their standard symbols, which you'll find in the aleks calculator.
Answers: 2

Chemistry, 23.06.2019 11:00
Which of the following reactions is endothermic? h2(g) + ½ o2(g) h2o(g), h = -57.82 kcal ½n2(g) + o2(g) + 8.1 kcal no2(g) ½ n2(g) + 3/2 h2(g) nh3(g) + 11.0 kcal c(diamond) + o2(g) co2, h = -94.50 kcal
Answers: 2
You know the right answer?
Questions


History, 10.10.2020 23:01


Mathematics, 10.10.2020 23:01

French, 10.10.2020 23:01

Mathematics, 10.10.2020 23:01

Advanced Placement (AP), 10.10.2020 23:01

English, 10.10.2020 23:01


Social Studies, 10.10.2020 23:01



Mathematics, 10.10.2020 23:01

Chemistry, 10.10.2020 23:01


Mathematics, 10.10.2020 23:01


Health, 10.10.2020 23:01

History, 10.10.2020 23:01

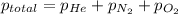
 = total pressure of gases = 752 mm Hg
= total pressure of gases = 752 mm Hg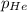 = partial pressure of Helium = 234 mm Hg
= partial pressure of Helium = 234 mm Hg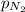 = partial pressure of nitrogen = 197 mm Hg
= partial pressure of nitrogen = 197 mm Hg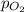 = partial pressure of oxygen = ?
= partial pressure of oxygen = ?



