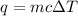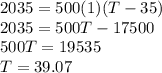If 2,035 cal of heat is added to a 500.0 g sample of water at 35.0°C, what is the final
temper...

Chemistry, 05.05.2020 10:45 maddie0533
If 2,035 cal of heat is added to a 500.0 g sample of water at 35.0°C, what is the final
temperature of the water?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:00
Agas in a balloon at constant pressure has a volume of 160 ml at -125*c. what is its volume at 29.0*c?
Answers: 1

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2
You know the right answer?
Questions

English, 23.04.2021 23:10

Mathematics, 23.04.2021 23:10

History, 23.04.2021 23:10







Computers and Technology, 23.04.2021 23:10




Mathematics, 23.04.2021 23:10

English, 23.04.2021 23:10

Mathematics, 23.04.2021 23:10