
Calculate Z and V for ethylene at 25°C and 12 bar by the following equations: (a) The truncated virial equation [Eq. (3.38)] with the following experimental values of virial coefficients: B = −140 cm3·mol−1 C = 7200 cm6·mol−2 (b) The truncated virial equation [Eq. (3.36)], with a value of B from the generalized Pitzer correlation [Eqs. (3.58)–(3.62)] (c) The Redlich/Kwong equation (d) The Soave/Redlich/Kwong equation (e) The Peng/Robinson equation

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2
You know the right answer?
Calculate Z and V for ethylene at 25°C and 12 bar by the following equations: (a) The truncated viri...
Questions

Mathematics, 29.10.2019 17:31


Chemistry, 29.10.2019 17:31


English, 29.10.2019 17:31


History, 29.10.2019 17:31

History, 29.10.2019 17:31




Mathematics, 29.10.2019 17:31

Mathematics, 29.10.2019 17:31

English, 29.10.2019 17:31




Advanced Placement (AP), 29.10.2019 17:31

History, 29.10.2019 17:31

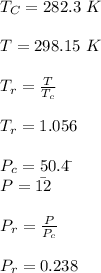
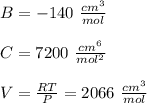
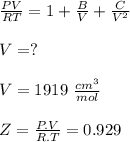
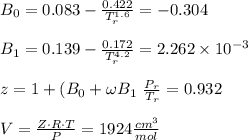
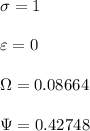
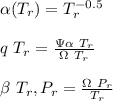
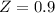
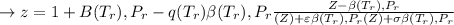
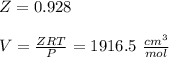
![\sigma =1\\\\\varepsilon = 0\\\\ \Omega= 0.08664\\\\ \Psi= 0.42748 \\\\\alpha (T_r) \omega =[ 1+ 0.480 + 1.574 \omega -0.176 \omega^2 (1-T_r^{\frac{1}{2}})]^2\\\\ \alpha (T_r)=\frac{\Psi \alpha (T_r) , \omega}{\Omega \ T_r}\\\\\beta (T_r), P_r=\frac{\omega\ P_r}{T_r}\\\\](/tpl/images/0640/3828/ecc5c.png)
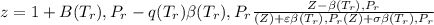
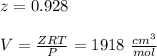
![\sigma =1+\sqrt{2}\\\\\varepsilon = 1-\sqrt{2}\\\\ \Omega= 0.07779\\\\ \Psi= 0.45724 \\\\\alpha (T_r) \omega =[ 1+ 0.37464 + 1.5422 \omega -0.26992 \omega^2 (1-T_r^{\frac{1}{2}})]^2\\\\ \alpha (T_r)=\frac{\Psi \alpha (T_r) , \omega}{\Omega \ T_r}\\\\\beta (T_r), P_r=\frac{\omega\ P_r}{T_r}\\\\](/tpl/images/0640/3828/e71f8.png)




