
Chemistry, 05.05.2020 18:46 jerrygentry3411
Explain what types of solutions are being combined. Use the terms titrant and analyte in your explanation. Justify your answer.
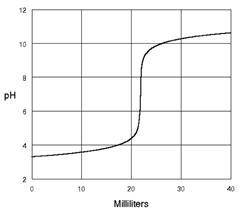

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 07:00
What effect does a decrease in temperature have on the overall rate of a chemical reaction? a decrease in temperature decreases . the reaction rate will
Answers: 1

Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2
You know the right answer?
Explain what types of solutions are being combined. Use the terms titrant and analyte in your explan...
Questions


Mathematics, 24.11.2020 18:30

Business, 24.11.2020 18:30

English, 24.11.2020 18:30

English, 24.11.2020 18:30

Chemistry, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30





History, 24.11.2020 18:30



Health, 24.11.2020 18:30


Biology, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30

Mathematics, 24.11.2020 18:30



