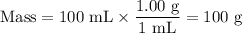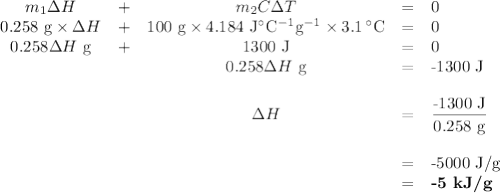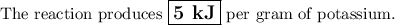Chemistry, 06.05.2020 03:42 pennygillbert
A 0.258 g piece of potassium solid is placed inot water inside a coffee cup calorimeter resulting in a vigorous reaction. assume a total volume of 100 ml for the resulting solution. the temperature of the solution changes from 22 to 25.1 due to the reaction. how much heat in kj is generated per gram of potassium for this reaction? assume the density of the solution after the reaction is the same as the density of water

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1

Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2

Chemistry, 22.06.2019 22:10
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1
You know the right answer?
A 0.258 g piece of potassium solid is placed inot water inside a coffee cup calorimeter resulting in...
Questions






History, 07.05.2020 13:01

Mathematics, 07.05.2020 13:01

English, 07.05.2020 13:57

Biology, 07.05.2020 13:57


History, 07.05.2020 13:57

History, 07.05.2020 13:57

Advanced Placement (AP), 07.05.2020 13:57

History, 07.05.2020 13:57




Chemistry, 07.05.2020 13:57


Chemistry, 07.05.2020 13:57