Chemistry, 06.05.2020 15:57 colexie7410
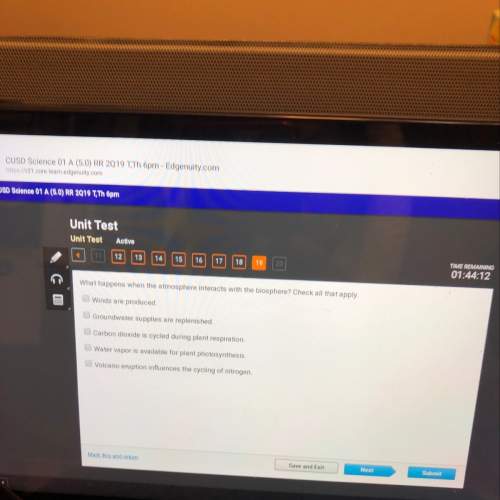
10. A basic solution of Al(OH)3, has a pH of 9.85, what is the [OH-] concentration of the aluminum
hydroxide solution? *

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

Chemistry, 23.06.2019 05:40
Convert a speed of 201 cm/s to units of inches per minute. also, show the unit analysis by dragging components into the unit‑factor slots.
Answers: 1

Chemistry, 23.06.2019 10:30
4al + 3o2 → 2al2o3 what does the "3" in front of o2 stand for? a) it indicates that there are 5 oxygen atoms after you add the coefficient and the subscript. b) it indicates that that there are are total of 6 oxygen atoms all bonded together as a single molecule. c) it indicates that there are 3 oxygen molecules chemically bonded to each other in the reaction. d) it indicates that there are 3 separate oxygen molecules in the reaction.
Answers: 2

You know the right answer?
10. A basic solution of Al(OH)3, has a pH of 9.85, what is the [OH-] concentration of the aluminum
Questions



History, 24.11.2020 19:30



Social Studies, 24.11.2020 19:30


English, 24.11.2020 19:30

Business, 24.11.2020 19:30

Computers and Technology, 24.11.2020 19:30

History, 24.11.2020 19:30

Mathematics, 24.11.2020 19:30


English, 24.11.2020 19:30



Mathematics, 24.11.2020 19:30

Physics, 24.11.2020 19:30

Mathematics, 24.11.2020 19:30

Social Studies, 24.11.2020 19:30

![[OH-]](/tpl/images/0648/6838/2930c.png) concentration of the aluminum hydroxide solution is
concentration of the aluminum hydroxide solution is 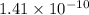
![pOH=-\log [OH^-]](/tpl/images/0648/6838/1fac1.png)
![9.85=-\log[OH^-]](/tpl/images/0648/6838/4631a.png)
![[OH^-]=10^{-9.85}=1.41\times 10^{-10}](/tpl/images/0648/6838/2535c.png)