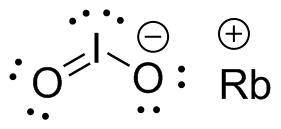
The RbIO₂ compound has a Lewis structure in IO₂⁻
Further Explanation
The Lewis formula is used to describe covalent bonds
Lewis structures have a central atom and a terminal atom
The central atom is an atom that is bound to 2 or more other atoms, while a terminal atom is bound to 1 other atom
In describing Lewis's structure the steps that can be taken are:
1. Count the number of valence electrons from atoms in a molecule
2. Give each bond a pair of electrons
3. The remaining electrons are given to the atomic terminal so that an octet is reached
4. If the central atom is not yet octet, free electrons are drawn to the central atom to form double bonds
In the RbIO₂ compound, the bond is an ionic bond that is Rb + and IO₂⁻ so that what Lewis can describe is the I and O bonds in IO₂⁻
Steps to draw the Lewis structure
1. Place atom I in the middle and O atom on the side
2. Draw the electrons that bind each of 2 pairs to form 2 bonds
3. Write down the remaining free electrons after subtracting the bonding electrons
4. The form of a double bond of one O atom with atom I, because there are electrons that have not been paired
From this it was found that atom I did not follow the octet rule because it binds 10 electrons, this is what is called the octet rule which was developed / expanded, whereas in general period 3 elements such as P, Br, I can have electrons exceeding 8 when they bind
Learn more
adding electron dots as needed h3c ss ch3
link
formal charge
link
ionic bonding
link
Keywords: Lewis structure, single-bonded, double-bonded, octet