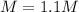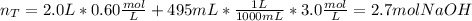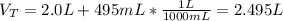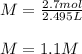What is the molarity of sodium hydroxide solution made by combining 2.0 L of 0.60
NaOH With 49...

Chemistry, 17.05.2020 04:57 deshundradenton482
What is the molarity of sodium hydroxide solution made by combining 2.0 L of 0.60
NaOH With 495 mL 3.0 M NaOH? Assume the volumes of the two solutions to be additive___M

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Which of the following best defines homeostasis? forming identical cells breaking down glucose maintaining stable internal conditions increasing an organism's temperature
Answers: 3

Chemistry, 22.06.2019 00:30
Used the balanced equation 2h2+ o2 - -> 2h2o. if you have 7.2 grams of o2 , how many grams of h2o can you produce ?
Answers: 2

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1
You know the right answer?
Questions


Biology, 23.02.2021 22:20


Mathematics, 23.02.2021 22:20


English, 23.02.2021 22:20


Engineering, 23.02.2021 22:20

Social Studies, 23.02.2021 22:20

Social Studies, 23.02.2021 22:20


Mathematics, 23.02.2021 22:20

Mathematics, 23.02.2021 22:20


Mathematics, 23.02.2021 22:20

Business, 23.02.2021 22:20

Mathematics, 23.02.2021 22:20

Mathematics, 23.02.2021 22:20


Mathematics, 23.02.2021 22:20