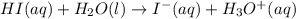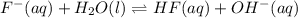Chemistry, 21.05.2020 00:07 Christyy9608
Identify the reactant that is a Brønsted−Lowry acid in the following reaction: HI(aq)+H2O(l)→I−(aq)+H3O+(aq) Express your answer as a chemical formula. nothing Request Answer Part B Identify the reactant that is a Brønsted−Lowry base in the following reaction: HI(aq)+H2O(l)→I−(aq)+H3O+(aq) Express your answer as a chemical formula. nothing Request Answer Part C Identify the reactant that is a Brønsted−Lowry acid in the following reaction: F−(aq)+H2O(l)⇌HF(aq)+OH−(aq) Express your answer as a chemical formula. nothing Request Answer Part D Identify the reactant that is a Brønsted−Lowry base in the following reaction: F−(aq)+H2O(l)⇌HF(aq)+OH−(aq) Express your answer as a chemical formula.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:10
Which statement describes both homogeneous mixtures and heterogeneous mixtures?
Answers: 1

Chemistry, 22.06.2019 02:30
Which compound contains both ionic and covalent bonds? a) hbr b)cbr4 c)nabr d) naoh
Answers: 2


Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1
You know the right answer?
Identify the reactant that is a Brønsted−Lowry acid in the following reaction: HI(aq)+H2O(l)→I−(aq)+...
Questions



English, 26.07.2019 00:30

Mathematics, 26.07.2019 00:30


Mathematics, 26.07.2019 00:30

English, 26.07.2019 00:30

Social Studies, 26.07.2019 00:30

Mathematics, 26.07.2019 00:30

History, 26.07.2019 00:30


History, 26.07.2019 00:30


Mathematics, 26.07.2019 00:30

Mathematics, 26.07.2019 00:30

Biology, 26.07.2019 00:30

Mathematics, 26.07.2019 00:30

Computers and Technology, 26.07.2019 00:30


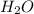 is Bronsted-Lowry base.
is Bronsted-Lowry base.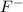 is Bronsted-Lowry base.
is Bronsted-Lowry base.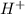 ).
).