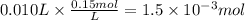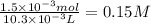Chemistry, 21.05.2020 05:00 cpratt2558
The student titrated 10 ml of standered 0.15 M HCl with his sodium hydroxide solution. When the titration reached the equivalence point, the student found that he had used
10.3 ml of Sodium hydroxide solution. Calculate the molarity of the sodium hydroxide solution.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3


Chemistry, 22.06.2019 17:20
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2
You know the right answer?
The student titrated 10 ml of standered 0.15 M HCl with his sodium hydroxide solution. When the titr...
Questions



Business, 16.02.2021 04:10







Mathematics, 16.02.2021 04:10



Biology, 16.02.2021 04:10

Advanced Placement (AP), 16.02.2021 04:10



Mathematics, 16.02.2021 04:10

Mathematics, 16.02.2021 04:10

Mathematics, 16.02.2021 04:10