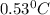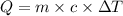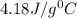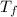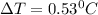Chemistry, 22.05.2020 04:08 potato1458
A 2.40 kg block of ice is heated with 5820 J of heat. The specific heat of water is 4.18 J•g^-1•C^-1. By how much will it’s temperature rise, assuming it does not melt?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
If you chip a tooth, most likely you will go to the dentist to have the missing material filled in. currently the material used to fill in teeth is a polymer that is flexible when put in, yet is hardened to the strength of a tooth after irradiation with blue light at a wavelength of 461 nm. what is the energy in joules for a photon of light at this wavelength?
Answers: 1

Chemistry, 22.06.2019 22:00
In order to complete this lab. you will need to be familiar with some common chemistry terms. complete the chemical change puzzle and list the relevant terms and their meaning below a.rectant b.product c.supernate
Answers: 3

Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

Chemistry, 22.06.2019 23:00
In which region is the substance in both the solid phase and the liquid phase? 1 2. 3 4 mark this and return save and exit next
Answers: 2
You know the right answer?
A 2.40 kg block of ice is heated with 5820 J of heat. The specific heat of water is 4.18 J•g^-1•C^-1...
Questions




Mathematics, 12.02.2020 06:04




History, 12.02.2020 06:04

Mathematics, 12.02.2020 06:04

Mathematics, 12.02.2020 06:04

Mathematics, 12.02.2020 06:04





History, 12.02.2020 06:18

Mathematics, 12.02.2020 06:18

Chemistry, 12.02.2020 06:18


Mathematics, 12.02.2020 06:18