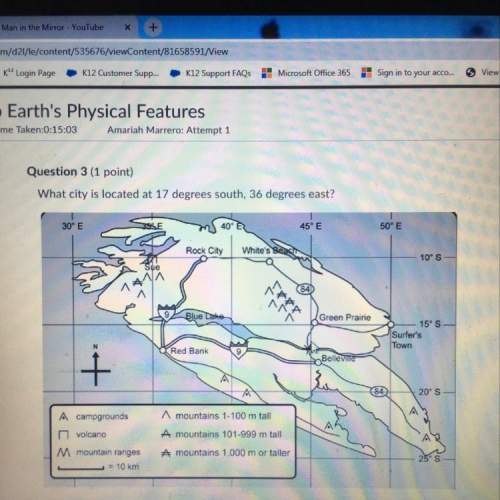
Consider the following reactions. (Note: (s) = solid, (l) = liquid, and (g) = gas.)
Mg(s) + ½O...

Consider the following reactions. (Note: (s) = solid, (l) = liquid, and (g) = gas.)
Mg(s) + ½O2(g) → MgO(s) + 146 kcal/mole
H2(g) + ½O2(g) → H2O(g), ΔH = -57.82 kcal/mole
What type of reaction is represented by the previous two examples?
A: exothermic
B: endothermic

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2

Chemistry, 22.06.2019 13:30
Mary is conducting an experiment on how pollution affects plant growth. how can she ensure that her data are reliable?
Answers: 3

Chemistry, 22.06.2019 16:30
How many grams of mgbr2 are needed to produce 75g or metal?
Answers: 1

Chemistry, 22.06.2019 20:30
Which states of matter have particles that move independently of one another with very little attraction?
Answers: 1
You know the right answer?
Questions

Chemistry, 21.08.2019 04:30


Mathematics, 21.08.2019 04:30

Physics, 21.08.2019 04:30





Geography, 21.08.2019 04:30

English, 21.08.2019 04:30

Social Studies, 21.08.2019 04:30



Arts, 21.08.2019 04:30

Mathematics, 21.08.2019 04:30

Social Studies, 21.08.2019 04:30




Mathematics, 21.08.2019 04:30