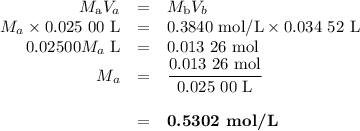Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

You know the right answer?
DUE TODAY EXTREMELY IMPORTANT PLEASE PLEASE PLEASE HELP ME ASAP CHEMISTRY
...
...
Questions



History, 08.07.2019 11:00




Mathematics, 08.07.2019 11:00





English, 08.07.2019 11:00



Mathematics, 08.07.2019 11:00


English, 08.07.2019 11:00

English, 08.07.2019 11:00

Mathematics, 08.07.2019 11:00