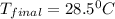Chemistry, 29.05.2020 20:00 rawaanasser12245
The specific heat of mercury is 0.138 J/g Co . If 452g of mercury at 85.0 Co are placed in 145g of water at 23.0 Co , what will be the final temperature for both the mercury and the water?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1


Chemistry, 22.06.2019 13:00
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3

Chemistry, 22.06.2019 19:40
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1
You know the right answer?
The specific heat of mercury is 0.138 J/g Co . If 452g of mercury at 85.0 Co are placed in 145g of w...
Questions

Mathematics, 28.09.2020 14:01



English, 28.09.2020 14:01




Mathematics, 28.09.2020 14:01

Mathematics, 28.09.2020 14:01


Mathematics, 28.09.2020 14:01




Social Studies, 28.09.2020 14:01

Mathematics, 28.09.2020 14:01

English, 28.09.2020 14:01

English, 28.09.2020 14:01


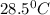
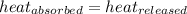
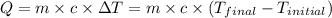
![m_1\times c_1\times (T_{final}-T_1)=-[m_2\times c_2\times (T_{final}-T_2)]](/tpl/images/0670/5209/09236.png) .................(1)
.................(1)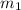 = mass of mercury = 425 g
= mass of mercury = 425 g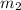 = mass of water = 145 g
= mass of water = 145 g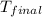 = final temperature = ?
= final temperature = ?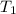 = temperature of mercury =
= temperature of mercury = 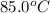
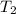 = temperature of water =
= temperature of water = 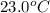
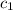 = specific heat of mercury =
= specific heat of mercury = 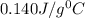
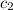 = specific heat of water=
= specific heat of water= 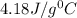
![-425\times 0.140\times (T_{final}-85.0)=[145\times 4.184\times (T_{final}-23.0)]](/tpl/images/0670/5209/c9617.png)