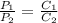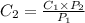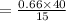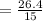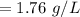Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3

Chemistry, 22.06.2019 07:00
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2


Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2
You know the right answer?
The solubility of a gas in water is 0.66 g/L at 15 kPa of pressure. What is the solubility when the...
Questions




Biology, 15.12.2019 16:31



History, 15.12.2019 16:31

English, 15.12.2019 16:31



Social Studies, 15.12.2019 16:31

History, 15.12.2019 16:31

Social Studies, 15.12.2019 16:31





Computers and Technology, 15.12.2019 16:31