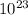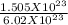A sample of sugar (C12H22O11) contains
1.505 × 1023 molecules of sugar. How many moles of suga...

Chemistry, 02.06.2020 22:00 chiarag305
A sample of sugar (C12H22O11) contains
1.505 × 1023 molecules of sugar. How many moles of sugar are present in the sample? Answer without doing any calculations.
0.25 mol
0.50 mol
1.00 mol
2.50 mol

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:50
Problem page gaseous ethane reacts with gaseous oxygen gas to produce gaseous carbon dioxide and gaseous water . if of water is produced from the reaction of of ethane and of oxygen gas, calculate the percent yield of water. be sure your answer has the correct number of significant digits in it.
Answers: 2

Chemistry, 21.06.2019 23:00
The drawing represents the movement of particles in a substance. what changes of state can this substance undergo
Answers: 1

Chemistry, 22.06.2019 03:20
Which type of substance ionizes partially and gives off hydrogen ions when dissolved in water? a. strong acid b. strong base c. weak acid d. weak base
Answers: 1

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3
You know the right answer?
Questions


Mathematics, 07.01.2021 08:30



Mathematics, 07.01.2021 08:30

Health, 07.01.2021 08:30

Mathematics, 07.01.2021 08:30






Chemistry, 07.01.2021 08:30

Mathematics, 07.01.2021 08:30


Chemistry, 07.01.2021 08:30


Mathematics, 07.01.2021 08:30