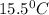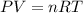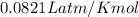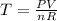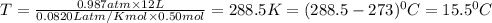Chemistry, 03.06.2020 09:57 muravyevaarina
Calculate the temperature of a 0.50 mol sample of a gas at 0.987 atm and a volume of 12 L.
-7 C
11 C
15.5 C
288 C

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 23:30
Rank the following four acids in order of increasing bronsted acidity : h2f+ , ch3oh, (ch3)2oh+ , ch3sh2+
Answers: 3

Chemistry, 23.06.2019 04:00
What are the names of these two interactions with cattle and how do they differ from each other
Answers: 3

Chemistry, 23.06.2019 05:30
Find the midpoint of a segment with endpoints of 4-3i and -2+7i
Answers: 2

Chemistry, 23.06.2019 08:00
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers
Answers: 1
You know the right answer?
Calculate the temperature of a 0.50 mol sample of a gas at 0.987 atm and a volume of 12 L.
Questions





Mathematics, 22.09.2020 22:01

Mathematics, 22.09.2020 22:01

History, 22.09.2020 22:01


Health, 22.09.2020 22:01

Computers and Technology, 22.09.2020 22:01

Mathematics, 22.09.2020 22:01



History, 22.09.2020 22:01


Spanish, 22.09.2020 22:01



Mathematics, 22.09.2020 22:01