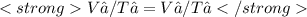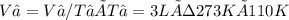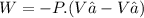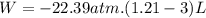Chemistry, 05.06.2020 00:00 SweetBriar
We now consider three moles of ideal gas at the same initial state (3.0 L at 273 K). This time, we will first perform an isobaric compression, then an isothermal expansion to bring the gas to the final state with the same volume of 3.0 L, and at temperature 110 K. Hint (a) How much work (in J) is done on the gas during the isobaric compression? Wcompression = J (b) How much work (in J) is done on the gas during the isothermal expansion? Wexpansion = J

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:40
During which time interval does the object travel approximately 10 meters
Answers: 3

Chemistry, 22.06.2019 03:00
Which step in naming unsaturated hydrocarbons is used for alkenes but not alkynes
Answers: 2

Chemistry, 22.06.2019 18:00
Which statement best describes the he properties of iconic compounds ?
Answers: 1

Chemistry, 22.06.2019 18:30
Which sample at stp has the same number of atoms as 18 liters of ne at stp
Answers: 1
You know the right answer?
We now consider three moles of ideal gas at the same initial state (3.0 L at 273 K). This time, we w...
Questions

Mathematics, 06.05.2020 05:09


Mathematics, 06.05.2020 05:09



Physics, 06.05.2020 05:09

Mathematics, 06.05.2020 05:09


Chemistry, 06.05.2020 05:09


Mathematics, 06.05.2020 05:09

English, 06.05.2020 05:09



Mathematics, 06.05.2020 05:09



Mathematics, 06.05.2020 05:09


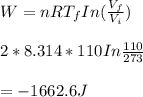

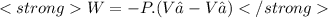
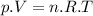
![p= [n.R.T] ÷ V = [3 mol × 0.082 (L.atm÷mol.K) × 273 K] ÷ 3 L](/tpl/images/0676/7490/eb257.png)