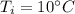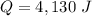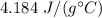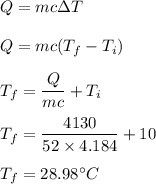Chemistry, 06.06.2020 04:00 zaniyastubbs9
A 52-gram sample of water that has an initial temperature of 10.0 °C absorbs 4,130 joules. If the specific heat of water is 4.184 J/(g °C), what is the final temperature of the water?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 23.06.2019 13:30
Did you mention that adding weight increased the pressure
Answers: 2

Chemistry, 23.06.2019 16:00
The table below shows a comparison of the different gas laws. some cells have been left blank. name variables constants equation boyle's law pressure, volume ? pv = k charles’s law volume, temperature ? v = kt ? temperature, pressure volume, moles of gas p = kt ? pressure, temperature, volume ? which are assumed to be constant while using the combined gas law? 1. pressure 2. number of moles 3. volume and moles of gas 4. pressure and temperature
Answers: 1
You know the right answer?
A 52-gram sample of water that has an initial temperature of 10.0 °C absorbs 4,130 joules. If the sp...
Questions

History, 08.04.2021 01:00


Mathematics, 08.04.2021 01:00

Mathematics, 08.04.2021 01:00




Mathematics, 08.04.2021 01:00

English, 08.04.2021 01:00



History, 08.04.2021 01:00

Physics, 08.04.2021 01:00

Chemistry, 08.04.2021 01:00

Mathematics, 08.04.2021 01:00

Mathematics, 08.04.2021 01:00

Engineering, 08.04.2021 01:00

Biology, 08.04.2021 01:00

Mathematics, 08.04.2021 01:00